Upcycling Polyolefins into Jet Fuel Components

A breakthrough method transforms HDPE waste into jet fuel components, boosting yield, quality, and sustainability.
Thermal conversion of plastic waste into fuel can replace fossil fuels while simultaneously mitigating environmental pollution. When using pyrolysis for polyolefins, catalysts can significantly reduce the energy required for thermal cracking. Zeolites are particularly beneficial for the production of hydrocarbons in the jet fuel range.
You can also read: Zeolites: A Mineral’s Role in Upcycling Plastic Waste.
Novel Method Converting HDPE into Jet Fuel
This method uses Fe/Beta catalysts, synthesized by impregnating Beta zeolites with a SiO2:Al2O3 ratio of 30. Decalin, a solvent and direct fuel blend component, reacts with Fe-zeolite catalysts during HDPE cracking. This produces oil with a high heating value (HHV) with significant potential as a jet fuel component.
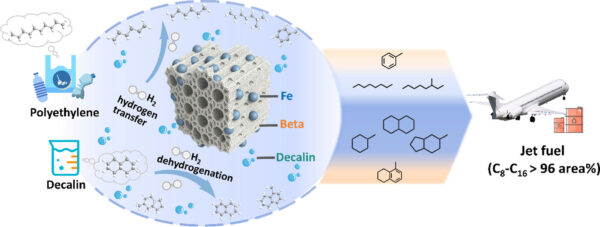
Researchers developed a methodology to obtain 84.0 wt% oil from HDPE. Figure courtesy of Efficient polyolefin plastic upcycling into jet fuel components over a Fe/Beta catalyst in decalin.
Catalyst Optimization
During development of this methodology, researchers evaluated a variety of catalysts with different Fe loadings. A Fe-loaded catalyst with a nominal 10 wt% Fe (10Fe/Beta) delivered optimal performance. Using this catalyst, researchers obtained an oil yield of 84.0 wt% under 300 °C for 90 minutes. This outperformed a commercial Beta zeolite catalyst alone, with an oil yield of 74.2 wt% primarily composed of gasoline-range hydrocarbons. Using the 10Fe/Beta catalyst, the resultant oil’s carbon number distribution was mainly concentrated in C8-C16. These carbon numbers indicate that the resulting product is suitable for jet fuel.
Solvent Thermal Liquefaction
Three stages comprise the thermal liquefication of polymers in solvent:
- Depolymerization/cracking
- Dissociation
- Recombination
During solvent thermal liquefaction (STL), HDPE melts and undergoes protonation and β-scission. Through electron transfer, Lewis acid sites activate C-H bonds to promote radical generation. As a hydrogen-donor solvent, decalin undergoes dehydrogenation and forms tetralin, releasing hydrogen from the catalyst surface. The STL process overcomes shortcomings in pyrolysis, such as secondary reactions like deep dehydrogenation and condensation. Dehydrogenative aromatization forms excessive aromatics, reducing coke formation. By incorporating decalin, STL becomes a feasible method for producing jet fuel.
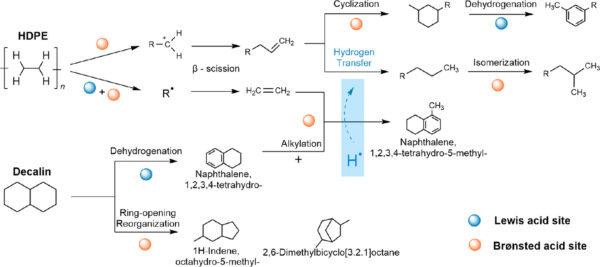
Decalin solvent increases the reaction pathways to transform HFPE to liquid fuels. Figure courtesy of Efficient polyolefin plastic upcycling into jet fuel components over a Fe/Beta catalyst in decalin.
Enhancing Yield of Fuels from HDPE
As a hydrogen donor solvent, decalin plays a critical role in this method, while researchers prepared the Fe/Beta catalyst using a simple impregnation approach. As a result, the catalytic reaction achieved an 84.0 wt% yield of liquid fuel, primarily composed of cycloalkanes and alkanes. In particular, the C8–C16 hydrocarbon fraction reached up to 96.9 area%, while delivering a high heating value of 44.5 MJ/kg. This performance is attributed to the uniformly dispersed Fe nanoparticles and the catalyst’s optimized acidity profile, which together enhance catalytic activity. Consequently, the process effectively suppresses the formation of aromatics and unsaturated hydrocarbons. Overall, this method improves both the yield and quality of liquid fuels suitable for jet fuel applications.

