Upcycling PET Through Artificial Photosynthesis

High-performance photocatalysts can upcycle post-consumer polyester under mild conditions.
Photocatalysis is an option for upcycling plastic waste under ambient conditions. Using solar energy, photocatalysts such as CdS, TiO2, and g-C3N4 induce redox reactions in plastic. Through the partial oxidation of plastics, these photocatalysts recover plastic’s carbon resources. This method offers a cost-effective alternative to traditional waste management, reducing carbon emissions while valorizing waste into high-value compounds.
You can also read: Bioinspired Hydrogels in Clean Energy and Hydrogen Generation.
Process of Photocatalysis
During photocatalysis, solar energy generates electron-hole pairs. Then, charge carriers migrate from the bulk to the photocatalyst surface. Redox reactions then occur on the photocatalyst’s surface. This process generates electrons, which can be used for water splitting or CO2 reduction to produce fuels. In turn, the resulting electron holes have applications in plastic valorization.
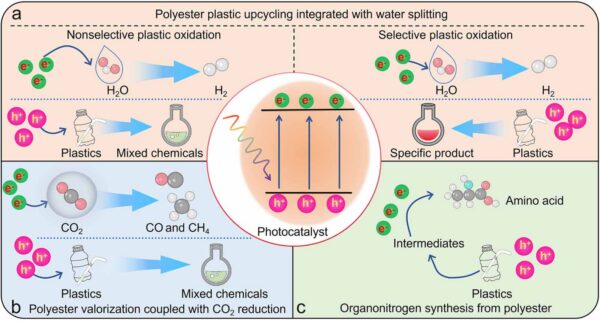
Photocatalysis has applications in polyester upcycling integrated with water splitting, valorization, and CO2 reduction, as well as in organonitrogen synthesis. Figure courtesy of Beyond mechanical recycling: artificial photosynthesis enables upcycling of polyester plastic into valuable chemicals.
Designing for Plastic Conversion
When designing photocatalysts, modifying the redox potential can enhance their effectiveness. Methods such as doping engineering and heterojunction constructions can achieve this.
- Element doping: This process introduces heteroatoms into the lattice of photocatalysts. This modifies the redox capacity of the photocatalyst by inducing changes in its energy band. Previous research investigated Cu doping of BiOBr, a photocatalyst, through a one-pot solvothermal method. This narrowed the band gap of BiOBr while tuning its redox capacity.
- Heterojunction constructions: Constructions such as type-II, Z-scheme, and S-scheme heterojunction are strategies for modifying redox potentials of photocatalysts.
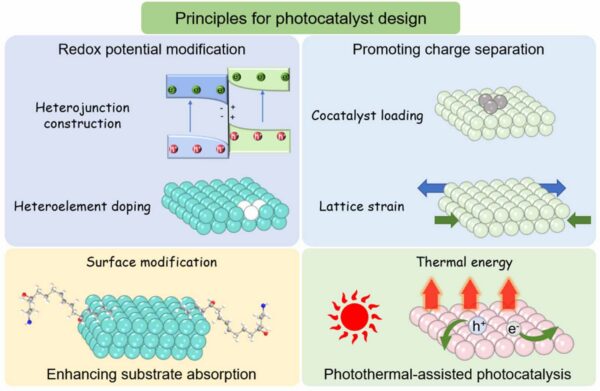
Surface modification, lattice strain, and thermal energy can also aid in photocatalyst optimization. Figure courtesy of Beyond mechanical recycling: artificial photosynthesis enables polyester plastic upcycling into valuable chemicals.
Artificial Photosynthesis for CO2 Adsorption
The overoxidation of polyester plastics generates CO2. Though the overabundance of CO2 leads to environmental issues, it is a crucial carbon resource. Artificial photosynthesis, or photocatalysis, requires sacrificial reagents to consume electron holes, increasing the cost of CO2 disposal. Alternatively, waste polyester can replace those sacrificial reagents during photooxidation. This leads to the conversion of both CO2 and waste polyester into reusable chemicals.
Research has shown that oxygen vacancy defect engineering in photocatalysts can enhance CO2 adsorption. Solvent and reaction temperature, as well as other parameters, can also affect the CO2 concentration in solution.
Photocatalyst Performance: TiO2 vs. g-C3N4
| Property | Titanium Dioxide (TiO2) | Graphitic Carbon Nitride (g-C3N4) |
| Band Gap (eV) | ~3.2 | ~2.7 |
| Primary Light Absorption | Ultraviolet (UV) | Visible Light |
| Stability | Excellent | Excellent |
| Main Advantage | Low cost, high abundance | Visible light activation |
| Main Limitation | Large band gap (requires UV) | Moderate recombination rate |
Harnessing the Power of Photocatalysis
Currently, challenges with photocatalysis limit its practicality for polyester upcycling. For example, only ultraviolet light and a small segment of visible light can induce polyester conversion. Since ultraviolet light makes up only ~5% of solar energy, developing photocatalysts that use near-infrared light is desired. Using simulations and artificial intelligence, researchers can predict how unknown photocatalysts will behave prior to experimentation.
Tandem catalytic systems, such as those that combine photocatalysis with electrocatalysis or thermal catalysis, can accelerate reforming of polyester. Introducing biocatalysis can further improve tandem systems.
