In Vivo Plastic Waste Upcycling

Advancements in biotechnology highlight how engineered microbial catalysts can recycle plastic waste in vivo.
Most current enzymatic approaches to managing plastic waste occur in vitro. These approaches include using enzymes to cleave plastic polymers, thereby releasing constituent monomers for repolymerization. Other bio-based approaches upcycle plastic waste into value-added compounds or use it as a carbon source for microorganisms. Novel research seeks to take this process in vivo. This entails using whole-cell microbial catalysts that feed directly on plastic waste, which could help address environmental challenges.
You can also read: Bio-Based Media for Micro- and Nanoplastics Removal.
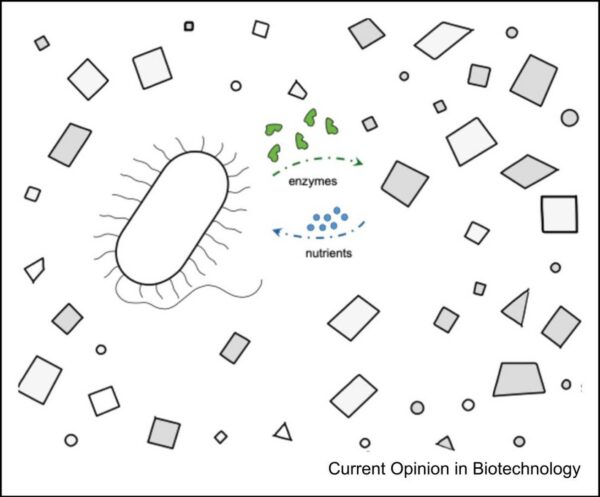
Advanced bio-based plastic waste management systems offer increased remediation potential but require further refinement. Figure courtesy of Engineering whole-cell catalysts to use plastic waste as a feedstock.
Whole-Cell Catalysts: Technical Challenges
Though whole-cell catalysts show promise for environmental remediation, challenges for researchers remain. Highly crystalline, tightly-packed polymer chains, for example, inhibit enzymatic hydrolysis. For polyethylene terephthalate (PET), this inhibition occurs when crystallinity exceeds 30%, which is common in post-consumer waste. Pretreatment methods, such as extrusion and mechanical shear, cryomilling, and solvent exposure can reduce crystallinity to overcome this.
Microplastics, however, are often highly crystalline. Conventional pretreatment strategies are not sufficient for microplastic treatment. Novel strategies, such as engineered microbes that secrete softening agents, may overcome this limitation.
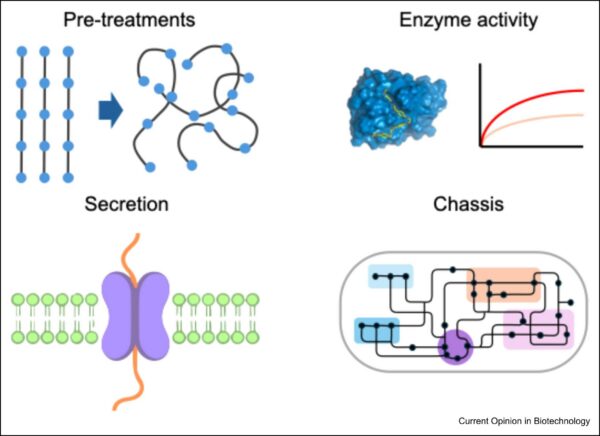
Designing pretreatments suitable for microplastics can enhance the effectiveness of bio-based waste treatment. Figure courtesy of Engineering whole-cell catalysts to use plastic waste as a feedstock.
Protein Engineering
Machine learning and artificial intelligence have contributed to recent advancements in enzyme optimization. Still, much of this research accounts only for in vitro conditions. Proteins that operate in vivo must do so under conditions compatible with cell viability. Therefore, the selection of usable enzymes remains limited. Because the local microenvironment affects enzyme kinetics, predictive models are essential to translate in vitro success to in vivo systems.
Secretion and Microbial Communities
For whole-cell plastic degradation, microbes must secrete enzymes to the extracellular medium. Most engineered species for plastic degradation are gram-negative bacteria, which have low secretion capabilities. Organisms that secret substantial amounts of protein, such as Bacillus subtilis and Pichia pastoris, could be adapted for plastic degradation. Other strategies for extracellular enzymatic activity include anchoring enzymes to the cell surface for direct contact with plastic substrates. Alternatively, engineered extracellular vesicles could carry enzymes into the environment.
Researchers have also explored the use of artificial microbial communities for plastic degradation. Combining organisms that specialize in different tasks may be key to avoiding challenges of a single-organism approach, like metabolic burden. Research has shown the effectiveness of this approach for PET hydrolysis and polyhydroxyalkanoate production using E. coli and P. putida.
Future Applications
Currently, most research on engineered microorganisms requires a laboratory environment. Engineering strains that survive in operational settings, colonize these environments, and express enzymes in situ would be an important milestone. As research continues to advance for whole-cell microbial degradation approaches, biosafety is an important consideration. Moving this technology out of the lab and into real-world applications will require regulation adherence and safety in the environment.
