The Regulatory Blind Spot in Plastic Design

The new EU 10/2011 and REACH mandates shift the focus of plastic compliance toward pigments and additives, affecting NIR recyclability and migration limits.
For the past decade, plastic regulation has focused on polymers. Policymakers targeted monomers of concern, recyclability targets, and resin-level bans. Environmental criteria entered slowly, often through voluntary programs and industry pledges. That focus is shifting.
You can also read: Transforming Black Plastic Recycling
Yet regulators are increasingly converging on a quieter vulnerability in plastic products. Additives, colorants, fillers, and effect pigments increasingly define whether a plastic product complies with the law. These materials give plastics their performance and appearance. They also introduce chemical, safety, and recycling risks that regulators can no longer ignore.
Regulation Is Catching Up with What Formulators Already Know
The shift is already visible in Europe. Commission Regulation (EU) No 10/2011 on plastic food contact materials, updated in January 2025, continues to expand its positive lists and migration limits, not only for base polymers but also for colorants, fillers, and functional additives.
National authorities push even further. Germany’s BfR Recommendation IX restricts acceptable colorant chemistries in consumer plastics, while Switzerland’s revised food contact framework and ink requirements create a stricter regime than the EU baseline.

Summary of updates under Switzerland’s RO 2024 755, tightening migration limits and group restrictions for additives in food contact plastics and packaging inks. Courtesy of SGS.
This divergence matters. A formulator may secure EU approval for an additive, yet lose access to Switzerland or specific applications such as toys. The European Commission’s proposed
Toy Safety Regulation, published in April 2025, illustrates the trend. The proposal imposes far tighter substance limits on products intended for children, including pigments and effect additives. Regulators now treat additives as core compliance drivers, not secondary inputs.
Microplastics and Recycling Redefine “Acceptable” Additives
Microplastics policy accelerates this shift. Commission Regulation (EU) 2023/2055 restricts intentionally added microplastics across many product categories. While cosmetics and coatings draw the most attention, the rule also affects polymer additives.
Pigments, fillers, and effect particles can shed, abrade, or fragment during use and recycling. Regulators increasingly evaluate these materials under REACH, even when designers never intended them to function as microplastics.
Research in Polymers and Waste Management shows how conventional carbon black disrupts near-infrared sorting The same studies demonstrate how hyperspectral imaging, rare-earth oxide markers, and taggant systems improve detection. Yet these solutions add new substances to formulations, which raises further safety and food-contact questions.
Industry initiatives such as HolyGrail 2.0 and guidance from the Association of Plastic Recyclers promote design-for-sorting principles. Regulators, however, have not aligned chemical safety, food contact, and microplastics frameworks for digital watermarks, tracers, or functional fillers. This gap creates a compliance gray zone exactly where circularity ambitions run highest.
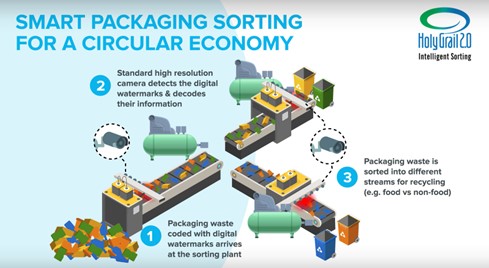
Digital watermarking enables high-precision sorting of packaging waste, improving recyclability across food and non-food plastic streams. Courtesy of Digital Watermarks.
A Patchwork Becomes a Strategic Risk
Across the Atlantic, the picture is no less complex. In the United States, FDA regulations govern colorants for polymers in food contact applications, supplemented by evolving Color Additive Inventories updated through the end of 2025.
Meanwhile, the Consumer Product Safety Commission continues to enforce strict limits on heavy metals such as lead in surface coatings, with implications for legacy pigments still used in niche industrial applications. The result is a patchwork: legally acceptable additives in one jurisdiction may be commercially unusable in another.
Suppliers have started to respond. UPM’s plans to launch a bio-based, NIR-detectable black pigment in late 2025. The product aims to combine recyclability, regulatory compliance, and sustainability claims in one solution.
These examples highlight a broader pressure. Formulators must now validate additives across multiple regulatory systems without unified guidance. Each decision carries legal, operational, and reputational risk and manufacturers must respond earlier in the design process. They need to align material choice, regulatory compliance, and recyclability from the start. The next phase of plastics regulation will not hinge on resin bans alone. It will hinge on the small substances that give plastics their color, texture, and function.
