Sustainable Plastics in Pharma: Insights from Dr. Beate Mueller-Tiemann

What it takes for sustainable plastics to work in pharma, with insights from Dr. Beate Mueller-Tiemann.
Sustainable plastics promise environmental improvement, but pharmaceutical production evaluates them differently. The question is not whether a material is better, but whether it behaves predictably every time it is used. That distinction shaped the discussion with Dr. Beate Mueller-Tiemann, whose work focuses on moving materials from development into validated manufacturing environments.
You can also read: AI Redefines Packaging with Faster Development Cycles.
Dr. Beate Mueller-Tiemann works across the full pharmaceutical value chain from discovery through commercial manufacturing. After leadership roles at Bayer and Sanofi, she joined Cytiva in 2023 to focus on turning innovation into dependable patient supply. Across biologics and synthetic molecules, she has seen the same barrier appear. A material may be promising, but unless its behaviour under production conditions is understood, it remains outside routine use.
Addressing the Start of the Value Chain
For companies producing plastics used in pharmaceutical applications, a practical question arises. As sustainable materials advance, what needs to change in how they approach development?
Dr. Mueller-Tiemann explains that developers operate at the very start of the pharmaceutical and medical product development process, which means downstream users rely on the information they provide. Decisions increasingly depend on measurable environmental data such as feedstock origin, carbon footprint, and impact on water, land, and biodiversity.
She also points to a recurring issue in development programs. Materials are sometimes created without an evidence-based end-of-life pathway. The problem then appears later in the product lifecycle. Sustainability requirements need to be embedded at the beginning of material and product design rather than addressed after development.
The same applies to performance expectations. Beyond regulatory compliance, pharmaceutical users require drop-in materials with recognized certifications, measurable emission reductions, and performance equal to or exceeding that of fossil-based alternatives. Manufacturing readiness and long-term supply security often matter more than cost.
Another overlooked area is the practical feasibility of end-of-life. Developers do not always evaluate whether materials can realistically be recycled, whether they enter biohazard waste streams, or how they fit global regulatory systems. Consistent Product Carbon Footprints and Digital Product Passports help provide the clarity required for adoption.
Why Promising Materials Stall in Evaluation
Even when sustainability metrics look favourable, adoption often stops at the evaluation stage. The obvious question becomes: where does the technical gap actually lie?
According to Dr. Mueller-Tiemann, the issue appears during real manufacturing use. Many materials lack robust comparative processing and performance data at a commercial scale. Without that information, companies cannot move forward, regardless of environmental benefit.
Circularity introduces another barrier. Current materials and use models frequently conflict with circular economy principles, thereby limiting reuse and recovery options.
For that reason, she considers sustainable plastics to still be in transition. Biobased polyethylene and polypropylene already reduce greenhouse gas emissions and represent a necessary first step. They do not yet solve end-of-life constraints. Long-term progress depends on combining sustainable sourcing, scalable manufacturing, and validated pathways for reuse, recycling, or regeneration.
Accelerating Qualification through Standardization
If the bottleneck is confidence, the next issue becomes qualification speed. Pharmaceutical validation processes are inherently cautious, so the industry is examining how to present material evidence more effectively.
Dr. Mueller-Tiemann points to standardized frameworks such as ISCC certification, Product Carbon Footprints, and Digital Product Passports aligned with pharmaceutical validation expectations. She also stresses collaboration across the value chain. Polymer producers, converters, pharmaceutical suppliers, regulators, and drug manufacturers must develop a shared understanding of material requirements. Generating evidence and coordination requires effort, but she considers it necessary to scale sustainable solutions responsibly.
Signs of Industrial Progress
Despite the constraints, development is moving faster than many expected. The natural question, then, is whether real optimism is justified.
Dr. Mueller-Tiemann points to several indicators. Monolayer film technologies, emerging materials such as PHA, and expanding take-back and recycling programs are advancing practical circularity. She also notes how biobased feedstocks have moved from niche concepts to mainstream pharmaceutical use within only a few years. The speed demonstrates what happens when innovation, data, and industrialization advance together.
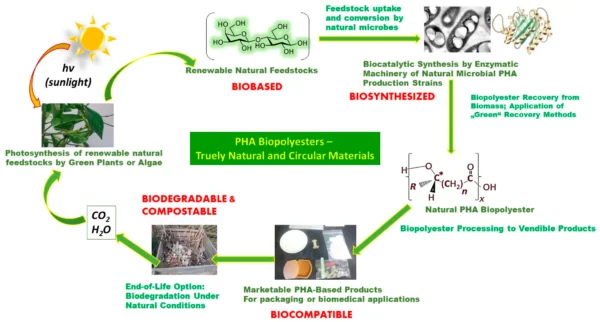
Circularity of natural PHA biopolyesters. Courtesy of Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural.
Her expectation is that sustainable plastics will succeed once they no longer need to be described as alternatives. They will simply be qualified materials selected through the same performance criteria as any other component in pharmaceutical manufacturing.
At ANTEC 2026, Dr. Beate Mueller-Tiemann will address the role of sustainable plastics in pharmaceutical manufacturing and the requirements for their responsible adoption.
